The single major responsibility of any Health Authority/Drug Regulatory Authority is to protect the health of its public.
Remember the term: PUBLIC HEALTH
That is also a reason why majority of drug regulatory authorities are nested under “Department of Health” within their Country.
For example:
- Food and Drug Administration (FDA), Drug Regulatory Authority in USA is nested under “The Department of Health and Human Services”.
- Medicines and Healthcare products Regulatory Agency (MHRA), the Drug Regulatory Authority in UK falls under “Department of Health and Social Care”
- Health Canada (HC), the Drug Regulatory Authority in Canada falls under “The Federal Ministry of Health”
- Therapeutics Good Administration (TGA) of Australia falls under Australian Government “Department of Health”
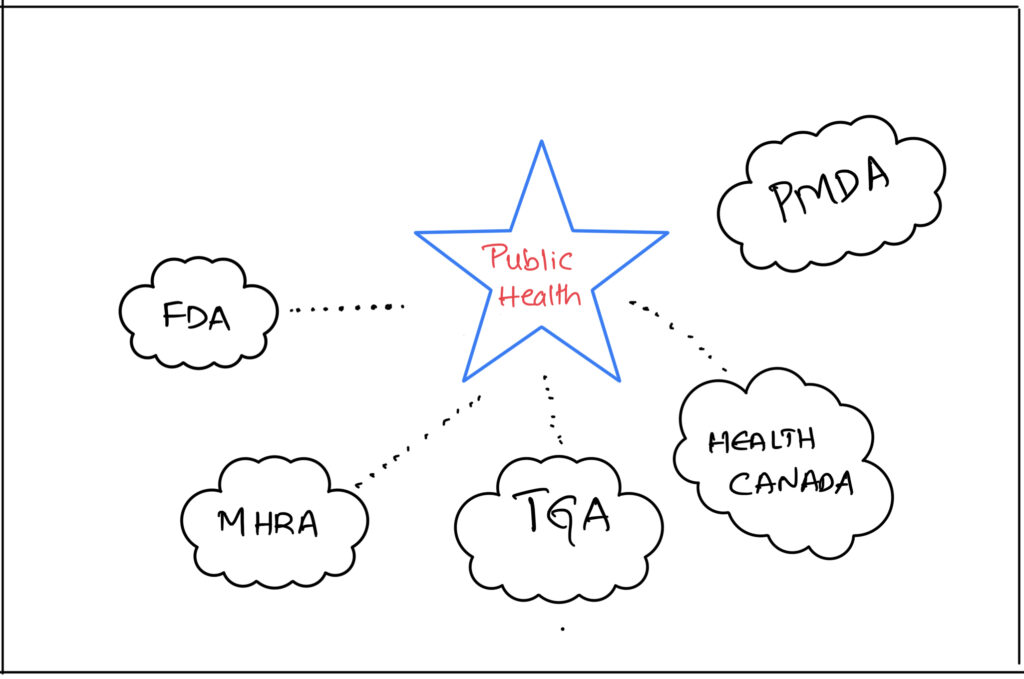
So, how do these authorities protect the public health?
Let us look into the mission statements of some of these agencies to get a better idea:
First Paragraph of United States FDA Mission says:
“The Food and Administration is responsible for protecting the public health by ensuring the safety, efficacy, and security of human and veterinary drugs, biological products, and medical devices; and by ensuring the safety of our nation’s food supply, cosmetics, and products that emit radiation”
Here is the mission statement for United Kingdom’s MHRA:
“The mission of the inspectorate is to protect the pubic health by making sure that medicines are available and are of right quality, applying appropriate standards of regulation”
Here is the mission statement from Republic of India’s Central Drug Standard Control Organization (CDSCO):
“To safeguard and enhance the public health by assuring the safety, efficacy and quality of drugs, cosmetics and medical devices”
Australia’s TGA describes the following in their regulatory performance framework:
“The TGA administers the Therapeutic Goods Act 1989, applying a risk management approach designed to ensure therapeutic goods supplied in Australia meet acceptable standards of quality, safety and efficacy (performance), when necessary.
You can see the commonality between these statements. Irrespective of processes they use to regulate drugs or cosmetics, their bulls eye is to protect the “Public Health”.
In my next post, I will write about how these mission statements fit into drug development.